This is a technical decision guide for engineers and procurement managers evaluating IrO₂ vs RuO₂ coated titanium anodes. If you already know MMO basics, jump directly to Section 4 — Application Decision Framework.
01Why the IrO₂ vs RuO₂ Decision Matters
When sourcing a Mixed Metal Oxide (MMO) titanium anode, most buyers focus on price per square meter or coating thickness. What they frequently overlook is that the choice of active oxide — iridium oxide (IrO₂) or ruthenium oxide (RuO₂) — determines whether the anode lasts 3 years or 10 years in your specific process environment. Choosing the wrong coating is one of the most common and costly mistakes in industrial electrochemistry.
Both IrO₂ and RuO₂ belong to the platinum-group metal (PGM) oxide family. Both are thermally decomposed onto a Grade 1 or Grade 2 titanium substrate and are available in mesh, plate, rod, or tubular form. But their electrochemical behaviour, stability windows, and cost profiles differ significantly — and those differences translate directly into operating cost and anode lifespan.
MMO (Mixed Metal Oxide) anodes — also called DSA® (Dimensionally Stable Anodes) — are titanium substrates coated with a thin layer of catalytic oxides applied by thermal decomposition. They replaced graphite and lead-alloy anodes in the 1970s because of their dimensional stability, superior electrocatalytic activity, and long service life. Read the full background in our Complete Guide to Titanium Anodes.
02The Chemistry Behind Each Coating
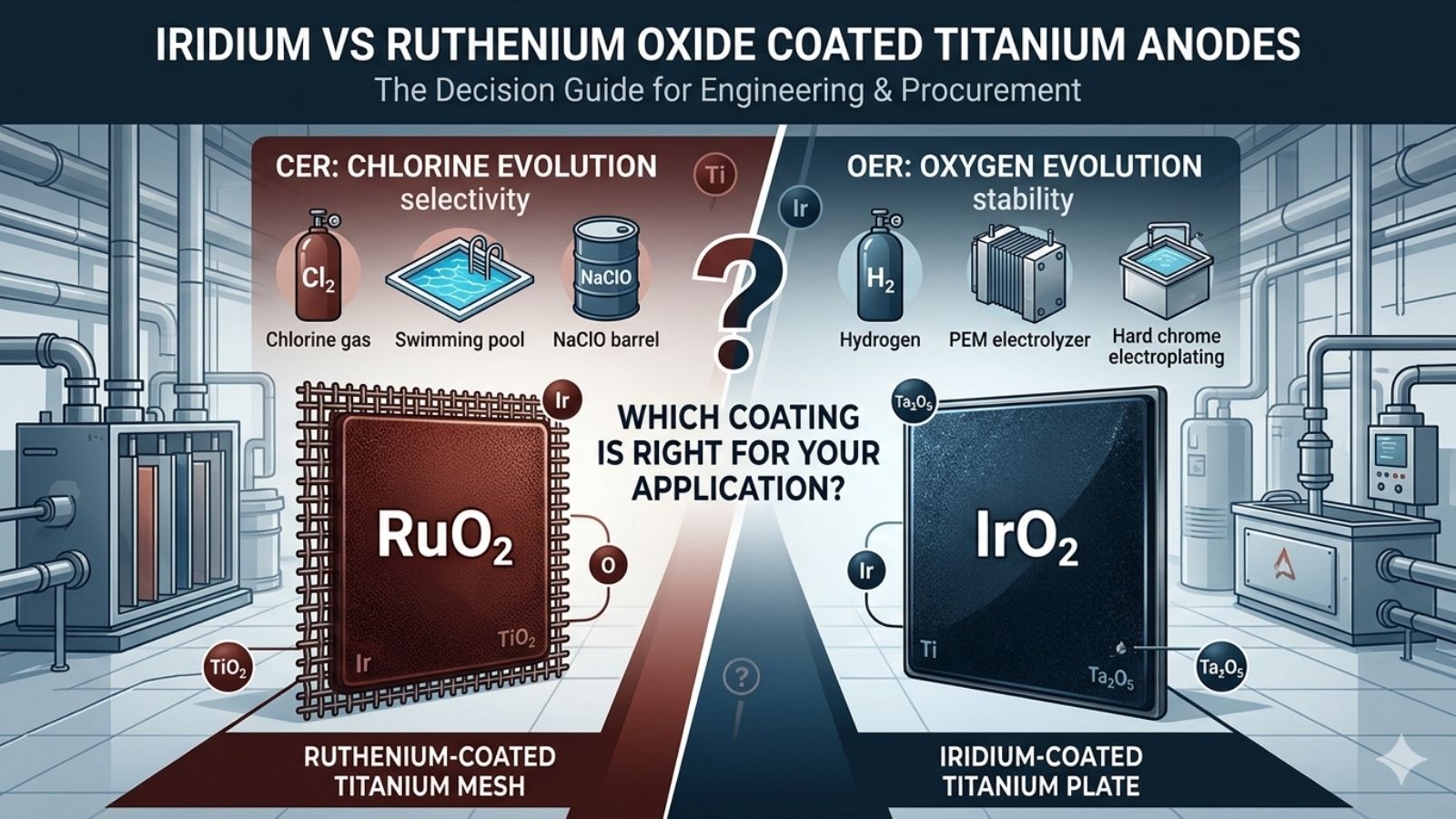
2.1 — Iridium Oxide (IrO₂)
IrO₂ is the dominant oxide used in applications where oxygen evolution (OER) is the primary anodic reaction. Its rutile-type crystal structure provides outstanding stability across the full pH range (0–14). The strong Ir–O bond energy is the key reason IrO₂ resists dissolution even under extreme anodic polarisation — a condition that rapidly destroys competing materials.
- Excellent stability under reverse current and voltage transients — critical for variable-load renewable energy systems
- Does not dissolve in strongly acidic OER environments, unlike RuO₂
- Compatible with strong NaOH (chlor-alkali cathode side) and concentrated H₂SO₄
- Standard coating for PEM water electrolysis — the only commercially proven oxide for acidic OER duty
In proton exchange membrane (PEM) electrolysers, the anode operates at pH < 1 and current densities up to 20,000 A/m². RuO₂ dissolves rapidly as RuO₄ under these conditions. IrO₂ is the only commercially proven oxide that survives thousands of operating hours in acidic OER — making it non-negotiable for green hydrogen production despite its higher cost.
→ See our detailed analysis: Titanium Anode Materials for PEM Water Electrolysis
2.2 — Ruthenium Oxide (RuO₂)
RuO₂ shares the same rutile crystal structure as IrO₂ but has a significantly lower chlorine evolution overpotential — around 1.36 V vs. SHE in NaCl solutions. This makes it the preferred active oxide for any process that needs to generate chlorine (Cl₂) or hypochlorite (ClO⁻) efficiently. In practice, JH Ti Anode supplies a ternary RuO₂+IrO₂+TiO₂ formulation: the IrO₂ fraction stabilises the coating, the TiO₂ filler optimises precious metal utilisation.
- Highest CER selectivity of any known coating — maximises Cl₂ yield while suppressing competing OER
- Lower electronic resistivity than IrO₂ — contributes to reduced overall cell voltage in brine systems
- Industrially proven in chlor-alkali for 50+ years; life exceeds 5 years at 1,000 A/m² in saturated NaCl
- Cost advantage: ruthenium spot price ~$16,000/kg vs iridium ~$175,000/kg (2025 reference)
03Head-to-Head Performance Comparison
| Parameter | IrO₂ Coating | RuO₂/IrO₂/TiO₂ Coating |
|---|---|---|
| Primary reaction | OER — O₂ evolution | CER — Cl₂ evolution |
| Target overpotential | ~1.52–1.56 V vs. SHE (OER) | ~1.36 V vs. SHE (CER in NaCl) |
| Max current density | Up to 18,000 A/m² | Up to 7,500 A/m² |
| Stability in strong acid (pH < 1) | Excellent — no dissolution | Moderate — Ru dissolves above ~1.6 V |
| Stability in alkaline media | Excellent | Excellent |
| Reverse current tolerance | High | Moderate — avoid prolonged reversal |
| Typical service life | 8–12 years (OER duty) | 5–8 years (CER duty, NaCl) |
| Precious metal cost (2025) | Higher (Ir ~$175,000/kg) | Lower (Ru ~$16,000/kg) |
| CER selectivity (Cl₂ yield) | Low — OER competes | High — optimised for CER |
| OEM custom loading (JH) | Ir: 10–60 g/m² on Gr1/Gr2 | Ru: 8–35 g/m² on Gr1/Gr2 |
Precious metal pricing referenced from Q1 2025 spot markets. Actual electrode system cost depends on coating area, loading specification, and configuration. Contact JH Ti Anode for project-specific pricing.
For a broader comparison including PbO₂ and platinum coatings alongside IrO₂ and RuO₂, see our technical report: Precious Metal Coatings vs. Standard Coatings — Comprehensive Performance Analysis.
04Application Decision Framework
The table below maps the most common industrial electrochemical processes to the recommended coating. Use it as your primary selection tool — your specific electrolyte chemistry, current density, and temperature may refine the recommendation further.
| Application | Recommended | Technical rationale |
|---|---|---|
| PEM water electrolysis (green H₂) | IrO₂ | Strongly acidic membrane (pH < 1); RuO₂ dissolves as RuO₄ under these conditions. IrO₂ is the only proven option. See: H₂ production anode page. |
| Chlor-alkali (Cl₂ + NaOH production) | RuO₂/IrO₂/TiO₂ | Saturated NaCl, 1,000 A/m², pH controlled near neutral. CER selectivity is critical for product purity and energy efficiency. |
| Sodium hypochlorite generation | RuO₂ or RuIr | Dilute brine CER duty. Ru-based coating maximises ClO⁻ yield at lowest energy cost. See: NaClO anode page. |
| Swimming pool salt chlorinator | RuO₂ or RuIr | Same CER chemistry as NaClO but lower duty cycle. See: pool salt chlorinator anode. |
| Cathodic protection — seawater | RuIr blend | Seawater Cl⁻ chemistry benefits from Ru CER selectivity; Ir stabilises the coating long-term (NACE TM0188). |
| Cathodic protection — soil / fresh water | IrO₂ + Ta₂O₅ | Pure OER environment; Ta₂O₅ stabiliser delivers 20–50 year certified service life. No chloride → no need for CER selectivity. |
| Copper electrowinning (hydrometallurgy) | IrO₂ (or IrTa) | Dilute H₂SO₄ at 200–400 A/m², pure OER duty. High acid stability essential; RuO₂ would dissolve within months. |
| PCB copper electroplating (acid bath) | RuIr blend | CuSO₄/H₂SO₄ bath — moderate acidity, OER/CER mix. RuIr blend balances stability and cost. See: PCB plating article. |
| Alkaline etching solution recovery (PCB) | IrO₂ + Ta₂O₅ | Alkaline NH₄Cl/NH₃ environment; iridium-tantalum specified for alkaline solution type per JH product data. |
| Precious metal plating (Au, Ag, Pd) | IrO₂ + Ta₂O₅ | Zero contamination tolerance. IrO₂ is inert with no ion leaching into the plating bath — critical for product quality. |
| Chromium electroplating (hard Cr) | IrO₂ | CrO₃/H₂SO₄ bath is highly oxidising. IrO₂ resists dissolution; avoids Pb contamination concerns of traditional Pb-Sn anodes. See: electroplating anode page. |
| Hospital / pharmaceutical wastewater | IrO₂-based MMO | No metal contamination of treated effluent. IrO₂ MMO meets regulatory discharge standards. See: hospital wastewater article. |
Not sure which row applies to your process? Submit a technical consultation request — our engineering team will recommend the correct coating and loading based on your operating parameters.
05The IrO₂ + Ta₂O₅ System: A Special Case
Tantalum pentoxide (Ta₂O₅) is not electrochemically active — it does not catalyse OER or CER. Its role is purely structural: Ta₂O₅ acts as a binder and spacer within the IrO₂ lattice, reducing sintering-induced grain growth during thermal decomposition and dramatically improving coating adhesion under mechanical stress.
The practical result is a harder, denser coating with superior resistance to reverse-current damage. This is why IrO₂+Ta₂O₅ is the standard formulation for cathodic protection ribbon and tubular anodes, where voltage reversal during lightning events or transformer faults is a real operational risk. JH Ti Anode supplies these to NACE TM0188-2008 certified performance standards.
Buried or submerged anodes with reversal risk · Service life targets above 20 years · Moderate, continuous (non-pulsed) current density · Non-chloride electrolyte (cathodic protection, not CER applications)
→ Product specifications including NACE ribbon and tubular data: Iridium-Coated Titanium Anode
06Cost Considerations: Price vs. Total Cost of Ownership
Iridium is one of the world's rarest elements — global annual production is approximately 7–8 tonnes. Scarcity drives its price to roughly 10× that of ruthenium. A higher Ir loading on a larger electrode area produces a meaningfully higher upfront cost for IrO₂ anodes. However, upfront material cost is often the wrong metric for procurement decisions.
What matters is the cost per ampere-hour of electrochemical work over the anode's full service life.
Figures are illustrative. Actual costs depend on precious metal spot pricing, electrode area, loading specification, and system downtime costs during replacement.
For a full framework including service life testing methodology and cost modelling across graphite vs titanium: Titanium Anodes vs Graphite Anodes: Service Life and Cost Analysis · Titanium Anode Lifespan Testing
07How Each Coating Fails — and How to Prevent It
IrO₂ — failure modes
RuO₂ — failure modes
For a deep dive into accelerated life testing (ALT) protocols and failure curve analysis: Titanium Anode Lifespan Testing: How to Accurately Predict Service Life.
08Specification Checklist for Your RFQ
When submitting a request for quotation (RFQ) to JH Ti Anode, providing the following information ensures you receive an accurate coating recommendation and competitive pricing from the first interaction.
Process parameters (mandatory)
Physical & commercial requirements
JH Ti Anode manufactures all coated anodes in-house at our Baoji facility. We offer full OEM/ODM services including custom shapes, non-standard dimensions, mixed-formulation coatings, and white-label supply for equipment integrators. All products are available with IrO₂, RuO₂, Pt, or PbO₂ coatings on Gr1/Gr2 titanium substrates.
09Quick Decision Guide
If you need a fast answer before diving into the technical detail, use these five-point filters:
Choose IrO₂ (or IrO₂ + Ta₂O₅) when:
- Your process evolves oxygen as the primary anodic reaction
- Your electrolyte is acidic (pH < 4) or strongly alkaline (pH > 12)
- You need current densities above 7,500 A/m²
- Application is PEM electrolysis, cathodic protection (non-seawater), copper electrowinning, or precious metal plating
- Service life target is 10+ years or long-duration infrastructure
Choose RuO₂/IrO₂/TiO₂ when:
- Your process generates chlorine or hypochlorite as the desired product
- Your electrolyte is chloride-based (NaCl, seawater, brine)
- Application is chlor-alkali, NaClO generator, pool salt chlorinator, or seawater disinfection
- Initial cost is a constraint and 5–8 year service life is acceptable
- You need the lowest possible cell voltage in a chloride electrolyte
For seawater applications where both CER and OER occur simultaneously, or for undivided cells with mixed electrolytes, the RuO₂/IrO₂/TiO₂ ternary blend is the standard recommendation — the IrO₂ component handles stability while RuO₂ maintains CER activity. Contact our engineering team for a free application review.
10Conclusion
IrO₂ and RuO₂ are not interchangeable — they are complementary solutions optimised for fundamentally different electrochemical reactions. The decision is not about which coating is "better" in absolute terms, but which is matched to your specific process chemistry, current regime, and lifecycle cost horizon.
The guiding principle is straightforward: if your anode needs to make chlorine efficiently and economically, RuO₂ is the answer. If your anode needs to evolve oxygen in a harsh, acidic environment without dissolving, IrO₂ is the only reliable choice. For mixed-duty or borderline cases, the RuO₂/IrO₂/TiO₂ ternary blend provides the best combination of CER selectivity and structural durability.
A poorly specified coating costs far more than a well-specified one — not in initial price, but in premature replacement, system downtime, and lost production. Taking 30 minutes to define your process parameters before requesting a quotation is the highest-ROI activity in the anode procurement process.
Explore JH Ti Anode Products
Related Articles
Ready to specify the right coating?
Our engineering team reviews your process parameters and recommends the exact IrO₂ or RuO₂ formulation, coating loading, and substrate configuration — backed by accelerated life test data.
Request a Free Consultation Download Product Brochure